A European skincare brand asked us to produce a multi-SKU run of airless pump bottles. Their previous supplier quoted a long lead time and required ordering each SKU separately in large batches. We delivered the full order in roughly half the time from a single consolidated production schedule. The difference was not faster machines. The difference was lean packaging production applied to every stage of the process, from mold changeover to final assembly.
Lean packaging production is a manufacturing methodology that eliminates non-value-added steps to reduce cost, shorten lead times, and improve consistency across every packaging run. For cosmetic brands sourcing from contract manufacturers, a supplier’s lean maturity directly determines whether your order ships on time, at the quoted cost, and at the quality your formulation requires.
What Lean Production Means in a Cosmetic Packaging Factory
Lean manufacturing originated with the Toyota Production System in the 1950s, but its application to cosmetic packaging involves specific challenges that general lean guides overlook. A cosmetic packaging factory handles injection molding, surface decoration (hot stamping, UV coating, silk screening), component assembly, and quality inspection across dozens of active SKUs. Each of those stages carries its own waste profile.
According to ISSSP, up to 95% of a typical packaging process may be non-value-added, with only product placement and labeling considered truly value-adding steps. That ratio makes lean a high-impact methodology for any factory that molds, decorates, and assembles cosmetic containers.
Lean production in cosmetics packaging is not about cutting corners. It is about identifying which activities add value from the customer’s perspective and systematically reducing everything else. When a brand orders 5,000 airless bottles with metallic hot stamping, value-adding steps include the injection molding cycle, the hot stamp application, pump assembly, and final QC. Non-value-adding steps include waiting between stations, excess inventory sitting on the floor, unnecessary material handling, and rework caused by preventable defects.
Oulete operates 20 injection molding machines with an annual capacity exceeding 20 million sets, and lean discipline is what keeps that volume moving at consistent quality. Without lean, high-volume production creates high-volume problems.
The Eight Wastes That Inflate Cosmetic Packaging Costs
The lean framework identifies eight categories of waste, known by the acronym DOWNTIME: Defects, Overproduction, Waiting, Non-utilized talent, Transportation, Inventory, Motion, and Extra processing. Each one shows up in specific ways inside a cosmetic packaging operation.
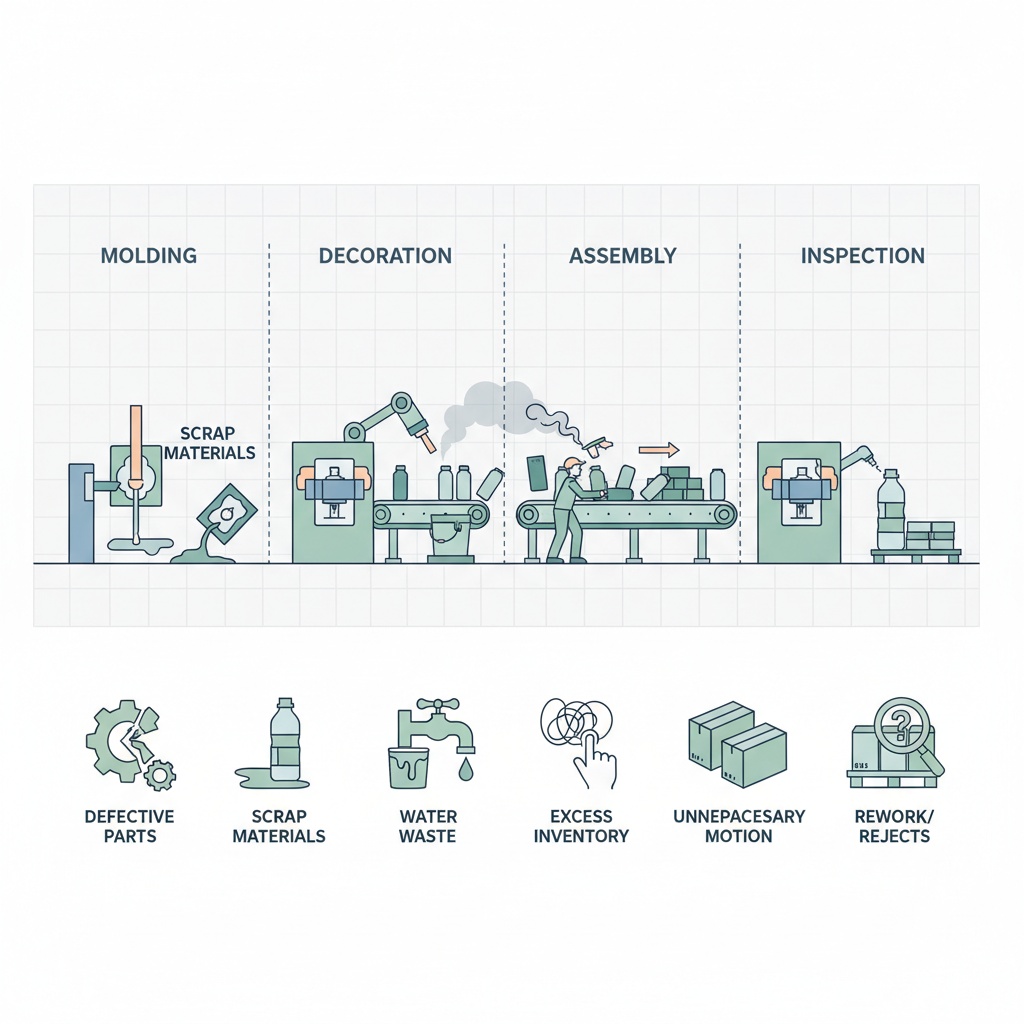
Defects in cosmetic packaging often surface during decoration. A misaligned hot stamp or uneven UV coat creates scrap that cannot be reworked because the base component has already been through an irreversible surface treatment. Overproduction happens when a factory molds more components than the order requires “just in case,” tying up material and storage space. Waiting occurs between injection molding and decoration when batch scheduling forces semi-finished parts to sit in staging areas for days.
Inventory waste is particularly damaging when PCR (Post-Consumer Recycled) materials are involved. PCR resin has more batch-to-batch variation than virgin material, so holding large PCR inventory increases the risk that stored material degrades or falls outside the processing window before it reaches the molding machine. Oulete’s PCR capability spans PP, PE, and PET at ratios from 10% to 50%, and lean incoming inspection protocols are what keep that material variation under control.
Motion waste in a packaging factory often hides in the assembly area, where operators reach for components stored too far from the workstation. Transportation waste appears when semi-finished parts travel between buildings or floors. Extra processing includes redundant inspection steps that exist because upstream defects were never addressed at their source. Non-utilized talent is the most commonly ignored waste: experienced operators who see problems daily but have no structured channel to report them.
Core Lean Tools Applied to Cosmetic Packaging Operations
Several lean tools translate directly to cosmetic packaging manufacturing. The critical tools for this industry are 5S, value stream mapping (VSM), Kaizen, SMED, and Kanban.
5S (Sort, Set in order, Shine, Standardize, Sustain) creates the foundation. On a decoration line handling hot stamping foils, UV coatings, and silk screen inks, 5S means every material has a labeled position, changeover tools are within arm’s reach, and cleaning schedules prevent contamination between color runs. This is not housekeeping for appearance. It is the operational backbone of GMP compliance.
Value stream mapping traces every step from raw resin delivery to finished goods shipment and measures the time each step takes. The goal is to calculate the ratio of value-adding time to total lead time. According to ISSSP, an emerging methodology called Green Integrated Value Stream Mapping (GIVSM) combines traditional VSM with carbon footprint assessment, which is relevant for cosmetic brands with ESG reporting commitments.
SMED (Single-Minute Exchange of Dies) reduces mold changeover time on injection molding machines. In a factory running multiple cosmetic container formats, the time spent switching from one mold to another is pure waste. Reducing changeover time from hours to minutes means more available production time per shift and the ability to run smaller batches economically.
Kanban uses visual signals to trigger production and material replenishment only when downstream consumption creates a need. This pull-based system prevents the overproduction and inventory waste that plague factories relying on forecast-driven push scheduling. Oulete’s ISO 9001-certified quality management system incorporates documented Kanban procedures for material flow control across all 20 injection molding machines.
Documented Results: What Lean Achieves in Packaging Manufacturing
The case for lean is built on measured outcomes, not theory. Multiple documented case studies show consistent patterns of improvement across packaging operations.
According to TXM Lean Solutions, a multinational packaging supplier operating four plants in China and one in Malaysia achieved lead times reduced by 28%, finished goods inventory down 32%, space utilization up 44%, and quality defects cut by 25% after lean implementation. These results came from a supplier serving electronics manufacturers, but the operational structure mirrors cosmetic packaging: multiple product families, decoration steps, and assembly operations.
According to ScienceDirect, a labeling and packaging plant study showed lean manufacturing improved lead time by 7.1%, inventory-to-cash conversion rate by 55%, and customer complaint rate by 83%. The complaint rate reduction is particularly relevant for cosmetic brands, where packaging defects visible to the end consumer damage brand perception.
According to Process Excellence Network, Anderson Packaging saved nearly $500,000 in waste-reduction benefits within less than one year of lean implementation in pharmaceutical packaging. Pharmaceutical packaging shares cosmetic packaging’s emphasis on contamination prevention and traceability, making this case study directly transferable.
| Metric | Before Lean | After Lean | Improvement |
|---|---|---|---|
| Lead time | Baseline | Reduced | 7% to 28% depending on complexity |
| Finished goods inventory | Excess stock common | Pull-based replenishment | Up to 32% reduction |
| Quality defects | Reactive detection | Source prevention | Up to 25% reduction |
| Space utilization | Underutilized floor area | Cellular layout | Up to 44% improvement |
| Customer complaints | Frequent | Systematic root cause analysis | Up to 83% reduction |
| Waste-related costs | Embedded in overhead | Identified and targeted | Up to $500K annual savings |
How Lean Production Lowers MOQ for Cosmetic Brands
One of the most practical benefits of lean for cosmetic brand buyers is its effect on minimum order quantities. High MOQs exist because traditional factories need large batch sizes to absorb changeover costs. When SMED reduces changeover time, the economic batch size shrinks, and the supplier can offer lower minimums without losing margin.
Oulete maintains an MOQ of 1,000 units, which is significantly lower than the industry norm for injection-molded cosmetic packaging. That low threshold is a direct result of lean changeover discipline. When switching between molds takes minutes rather than hours, running a 1,000-unit order becomes viable because the changeover cost per unit stays low.
This matters most for indie brands and startups that need to test market response before committing to large production volumes. A lean supplier enables smaller initial orders, faster reorders, and the ability to carry more SKUs without proportionally increasing packaging costs. The lean concept of “flow” replaces the traditional concept of “batch and queue,” which means orders move through the factory continuously rather than waiting in staging areas between departments.
According to Manifestly Checklists, over 70% of manufacturers that adopted lean in 2024 reported approximately 15% increases in operational efficiency, with lean factories typically using 10 to 25% less energy and producing up to 40% less scrap. Lower scrap rates directly support lower MOQs because the factory does not need to build excess units to compensate for expected defect losses.
Lean and GMP: Why ISO 22716 Aligns With Lean Principles
Cosmetic brands selling into regulated markets (EU under Regulation 1223/2009, US under MoCRA 2023) need suppliers that comply with ISO 22716, the international GMP standard for cosmetics. What many brands do not realize is that lean manufacturing and GMP share the same structural requirements.
ISO 22716 mandates documented processes, controlled production environments, traceability, and continuous improvement. Every one of those requirements is a lean principle. 5S creates the controlled environment. Standard work documentation fulfills the process documentation mandate. Kanban and pull systems create the traceability that GMP auditors look for. Kaizen provides the continuous improvement evidence.
Oulete holds ISO 9001, CE, SGS, and GMP certifications. Lean is not a separate initiative from our compliance program. Lean is the operational system that makes compliance sustainable rather than a burden. Factories that treat GMP as a paperwork exercise struggle during audits. Factories that run lean treat audit requirements as a natural output of how they already operate.
For brands evaluating suppliers, this alignment matters. A factory with genuine lean practices will have cleaner production areas, faster changeovers, lower defect rates, and better documentation than a factory that simply invested in newer equipment. Multi-cavity molding and automated assembly lines deliver throughput gains, but lean is what connects those capabilities into a reliable, repeatable system.
How to Tell Whether Your Supplier Is Truly Lean
Many packaging suppliers claim lean capabilities. Fewer can demonstrate them. When evaluating a cosmetic packaging manufacturer, these indicators separate genuine lean operations from marketing language.
Ask to see the factory’s value stream map. A lean factory will have a current-state VSM posted visibly and a future-state VSM showing planned improvements. Ask about changeover times on injection molding machines and decoration equipment. A genuinely lean factory tracks these numbers and can show you the trend over time.
Look at the production floor during your visit. Lean factories have minimal work-in-process inventory between stations, clearly marked material flow paths, and visual management boards showing daily production targets versus actuals. If you see large piles of semi-finished parts between injection molding and decoration, that factory is running batch-and-queue, not lean.
Ask about the suggestion system. Lean factories track employee improvement suggestions per month and implementation rates. Ask how they handle defects. A lean factory uses root cause analysis (the “5 Whys” method) and documents corrective actions. A non-lean factory replaces the defective part and moves on.
Finally, check certifications. ISO 9001 certification requires documented continuous improvement procedures. GMP compliance (ISO 22716) requires controlled production environments. These are not guarantees of lean maturity, but their absence is a reliable indicator that lean claims are superficial.
Frequently Asked Questions
What is lean packaging production?
Lean packaging production is a manufacturing methodology that systematically eliminates waste from every stage of the packaging process, including molding, decoration, assembly, and inspection. The approach reduces cost, shortens lead times, and improves quality consistency by focusing resources exclusively on activities that create value for the customer.
What are the 8 wastes in lean manufacturing applied to packaging?
The eight wastes follow the DOWNTIME acronym: Defects, Overproduction, Waiting, Non-utilized talent, Transportation, Inventory, Motion, and Extra processing. In cosmetic packaging, defects during decoration and waiting between production stages are typically the largest waste categories.
How does lean reduce packaging costs?
Lean reduces packaging costs by eliminating scrap, reducing inventory carrying costs, shortening lead times (which reduces working capital), and improving equipment utilization through faster changeovers. According to documented case studies, lean factories produce up to 40% less scrap and use 10 to 25% less energy than conventional operations.
What lean tools are most relevant for cosmetic packaging factories?
The five most relevant tools are 5S (workplace organization), value stream mapping (process analysis), SMED (changeover reduction), Kanban (pull-based scheduling), and Kaizen (continuous improvement events). Each addresses specific waste patterns found in injection molding, decoration, and assembly operations.
Can lean manufacturing help lower MOQs?
Lean directly enables lower minimum order quantities by reducing changeover costs per unit. When SMED compresses mold changeover time, the factory can economically produce smaller batches. This is why lean-mature suppliers can offer MOQs as low as 1,000 units for injection-molded cosmetic packaging.
How does ISO 22716 relate to lean production in cosmetics?
ISO 22716 (GMP for cosmetics) requires documented processes, controlled environments, traceability, and continuous improvement. These requirements overlap almost entirely with lean principles such as 5S, standard work, Kanban, and Kaizen. A lean factory meets most ISO 22716 requirements as a natural output of its operating system.
What results can a cosmetic brand expect when their supplier uses lean?
Documented results from packaging industry lean implementations include lead time reductions of 7% to 28%, inventory reductions up to 32%, quality defect reductions up to 25%, and customer complaint reductions up to 83%. Actual results vary based on the factory’s starting maturity level and the scope of implementation.
What is value stream mapping in packaging production?
Value stream mapping is a diagnostic tool that traces every step in the production process, from raw material receipt to finished goods shipment, measuring time, inventory, and information flow at each stage. The map reveals the ratio of value-adding time to total lead time and identifies the specific steps where waste concentrates.


