Last year, a U.S.-based indie skincare brand shipped a full production run of retinol cream to Amazon’s U.S. fulfillment centers. Customs and Border Protection flagged the entire shipment. The reason was not the formula. The packaging labels listed ingredients in alphabetical order instead of descending predominance, and the responsible person’s contact information was missing. The brand lost three months of shelf time and had to re-sleeve every unit at a domestic warehouse. FDA cosmetic packaging compliance requires meeting labeling, ingredient disclosure, and facility registration rules set by the FD&C Act and MoCRA 2022, all of which apply equally to domestically made and imported cosmetic packaging.
That situation is not unusual. According to Elchemy, approximately 48% of companies selling imported cosmetics at America’s top 25 retailers were non-compliant with FDA cosmetics regulations as of recent analysis. The packaging itself is often the failure point, not the formula inside it.
What FDA Cosmetic Packaging Compliance Actually Requires
The FDA does not approve cosmetics before they reach store shelves. Unlike drugs, there is no pre-market approval process for cosmetic products. According to the FDA’s own guidance, the only exception is color additives, which require FDA pre-market approval before they can be used in any cosmetic product.

FDA cosmetic packaging compliance is the set of labeling, safety substantiation, and registration obligations that govern how a cosmetic product is packaged, labeled, and tracked in the U.S. market. This compliance framework rests on three federal instruments: the Federal Food, Drug, and Cosmetic Act (FD&C Act) of 1938, the Fair Packaging and Labeling Act (FPLA), and the Modernization of Cosmetics Regulation Act (MoCRA) signed into law in December 2022.
For packaging manufacturers and brand owners sourcing from overseas, the practical consequence is straightforward. Your packaging must carry specific information, in specific locations, at specific type sizes. The product inside must be registered in a federal database. And the entity responsible for that product must be identifiable on every unit sold.
Oulete operates as a GMP-certified cosmetic packaging manufacturer with ISO 9001, CE, and SGS certifications. When brands ask us to produce packaging for the U.S. market, the compliance conversation starts before the first mold is cut. The physical design of the bottle or tube determines whether all required label elements can fit within FDA’s rules for the Principal Display Panel and the information panel.
The Three-Layer Regulatory Framework: FD&C Act, FPLA, and MoCRA
The FD&C Act has governed cosmetic safety and labeling since 1938. Under this law, cosmetic packaging must not be deceptive in form, fill, or shape. A container that appears to hold more product than it actually contains violates federal law. According to the FDA Cosmetics Labeling Guide, the FPLA adds specific requirements: net quantity of contents on the Principal Display Panel, product identity, and the name and address of the manufacturer or distributor.
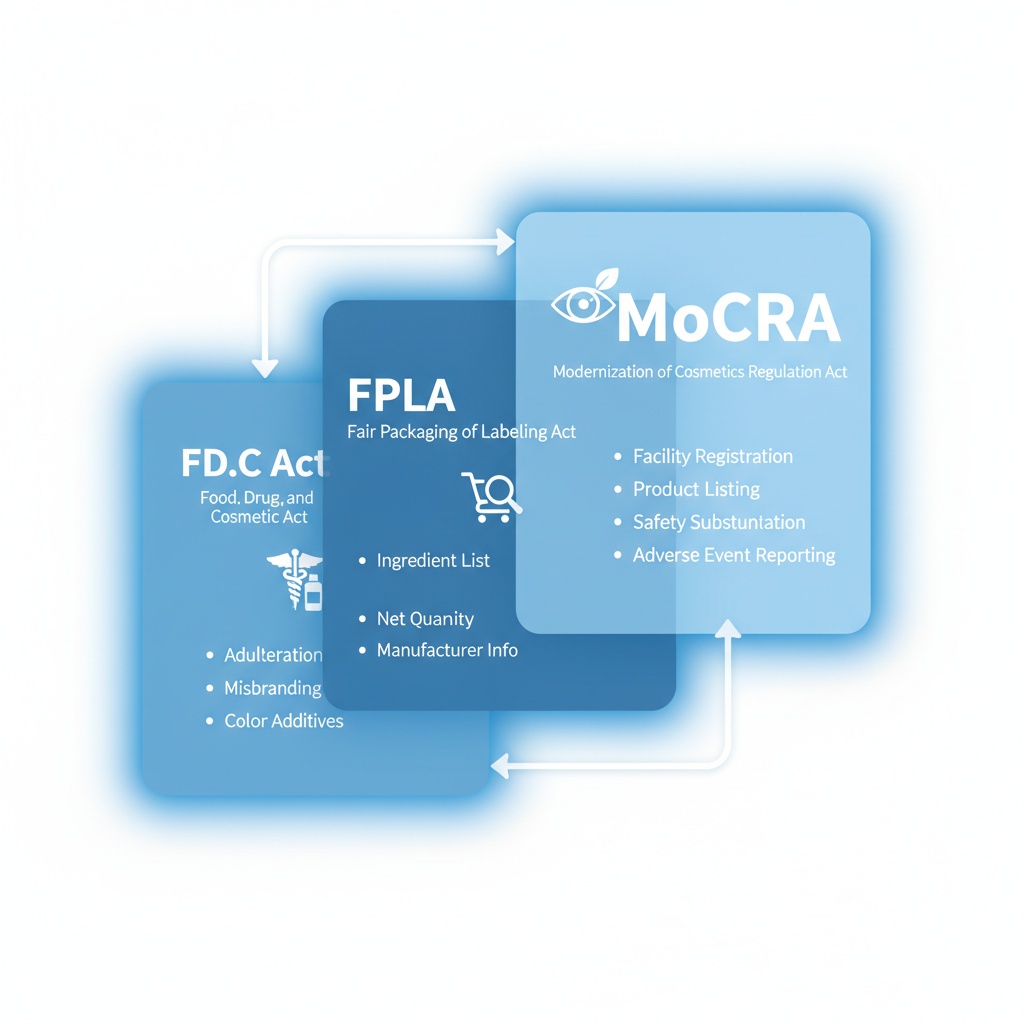
MoCRA is the Modernization of Cosmetics Regulation Act, a landmark 2022 law that represents the first major overhaul of U.S. cosmetics regulation since 1938. MoCRA directly affects every packaging supplier whose products are used in the U.S. market by introducing mandatory facility registration, product listing, adverse event reporting, and forthcoming Good Manufacturing Practice (cGMP) requirements.
MoCRA rolled out on a phased timeline. Professional-use-only products were required to carry label statements by December 29, 2023. The responsible person contact information requirement took effect December 29, 2024. According to Morgan Lewis, the mandatory cGMP rule for cosmetics was due December 29, 2025, but the FDA delayed finalizing the GMP rule and enforcement timeline beyond that date.
| Regulatory Layer | Enacted | Key Packaging Requirement | Enforced By |
|---|---|---|---|
| FD&C Act | 1938 | No adulterated or misbranded products; no deceptive packaging | FDA |
| FPLA | 1967 | Net quantity, product identity, manufacturer info on PDP | FDA / FTC |
| 21 CFR Part 701 | Ongoing | Label placement, type size, INCI ingredient order, language | FDA |
| 21 CFR Part 740 | Ongoing | Product-specific warning statements | FDA |
| MoCRA | 2022 | Facility registration, product listing, responsible person info, adverse event reporting | FDA |
On our production floor, we track these layers because each one has a physical implication for the packaging. The FD&C Act affects container shape and fill level. The FPLA and 21 CFR Part 701 dictate how much printable surface area a bottle needs. MoCRA added a new line of text (responsible person contact info) that must now fit on labels that were already designed before December 2024.
What Must Appear on Your Cosmetic Package Label
Every cosmetic sold in the United States must display specific information in specific locations. The Principal Display Panel (PDP) is the portion of the label most likely to be seen by the consumer at the point of purchase. According to the FDA Cosmetics Labeling Guide, the PDP must carry the product identity (what the product is) and the net quantity of contents.
The information panel is the label area immediately to the right of the PDP as the consumer faces the package. This panel must display the full ingredient declaration in descending order of predominance, the name and place of business of the manufacturer, packer, or distributor, and any required warning statements. Since December 29, 2024, MoCRA also requires the responsible person’s domestic contact information (U.S. address, phone number, or electronic contact) on this panel.
Ingredient names must follow INCI (International Nomenclature of Cosmetic Ingredients) nomenclature. According to ArtworkFlow, all ingredients present at more than 1% must appear in descending order by weight. Ingredients at or below 1% can be listed in any order after the above-1% ingredients.
Under 21 CFR Part 701, type size must be prominent and conspicuous, with adequate contrast against the background. The required minimum type size depends on the PDP area. This rule directly affects packaging design decisions. A 15 mL airless bottle has far less printable surface area than a 200 mL lotion bottle, yet both must carry the same mandatory information.
Oulete’s in-house decoration capability, including silk screening, hot stamping, and label application, functions as a compliance control point. When we print directly on the packaging, the label is permanently integrated with the container. This eliminates the risk of label peeling, misalignment, or application to the wrong SKU during third-party assembly.
Warning Labels and Product-Specific Requirements
Not all cosmetics carry the same warnings. The FDA requires product-specific warning statements under 21 CFR Part 740 for several product categories.

Aerosol cosmetics must include flammability and misuse warnings. Products sold in pressurized containers require statements about intentional inhalation and keeping the product away from heat sources. Tanning products without sunscreen must warn consumers that the product does not protect against sunburn. Feminine deodorant sprays require a specific caution statement about external use only.
Products with unsubstantiated safety must carry the statement: “WARNING: The safety of this product has not been determined.” This applies when a manufacturer has not performed adequate testing to substantiate the product’s safety for its intended use. MoCRA’s December 2023 deadline also added a new requirement: products intended for professional use only must state “FOR PROFESSIONAL USE ONLY” on the label.
Testing reveals that the warning statement requirement catches many brands off guard during packaging design. Warning text consumes label space. When a brand finalizes a label design without accounting for warnings, the only fix is either reducing other text sizes (which may violate minimum type size rules) or redesigning the container to provide more printable surface area.
Oulete manufactures airless pump bottles and tubes across a range of formats for the cosmetic market. When brands ask about compliance for aerosol or spray products, we advise them to map out every required text element before committing to a container size. A PETG bottle with a flat panel offers more usable label space than a cylindrical container of the same volume.
MoCRA Facility Registration and Product Listing
MoCRA introduced two entirely new obligations for the cosmetics industry: facility registration and product listing. According to Wiley Law, every facility that manufactures or processes cosmetics for the U.S. market must register with the FDA. Facility registration must be renewed biennially.

Product listing is the companion requirement. Every cosmetic product sold in the U.S. must be listed in the FDA’s database, with annual updates. According to Elchemy, as of January 2025, the FDA’s cosmetics database shows 9,528 unique active facility registrations and 589,762 unique active product listings, reflecting full MoCRA enforcement.
Small businesses with average gross annual U.S. cosmetic sales under $1 million are exempt from facility registration and product listing. According to Triage Health Law, this exemption does not apply to companies that make products intended for use in the area of the eye, injected products, or products intended for internal use.
Adverse event reporting is another MoCRA mandate. Serious adverse events (consumer reports of death, life-threatening experience, hospitalization, or significant disability) must be submitted to the FDA within 15 business days of receipt. This reporting obligation falls on the responsible person identified on the product label.
| MoCRA Requirement | Effective Date | Who Must Comply | Exemptions |
|---|---|---|---|
| Professional-use-only label statement | Dec 29, 2023 | All pro-use product sellers | None |
| Responsible person contact on label | Dec 29, 2024 | All cosmetic sellers | None |
| Facility registration | Phased (2024) | Manufacturers, processors | Small business (<$1M, with exceptions) |
| Product listing | Phased (2024) | All cosmetic sellers | Small business (<$1M, with exceptions) |
| Adverse event reporting | Ongoing | Responsible person | None |
| cGMP | Delayed past Dec 2025 | Manufacturers | Pending FDA rulemaking |
For brands sourcing packaging from China, the registration and listing obligations fall on the U.S.-based responsible person, not the overseas packaging manufacturer. But the packaging manufacturer’s quality systems directly support compliance. Oulete’s GMP certification aligns with the cGMP expectations that MoCRA will eventually enforce, giving our brand partners documented evidence of manufacturing controls.
Packaging Material Safety and Import Compliance
The FDA does not certify or approve packaging materials (plastics, glass, metals) specifically for cosmetics. However, the packaging must not contribute to the adulteration of the product inside. If a container leaches chemicals into the formula, or if the material degrades and contaminates the product, that product becomes adulterated under the FD&C Act.
Migration and leaching testing is the manufacturer’s responsibility. Brands sourcing PCR sustainable packaging or any recycled material must verify that the recycled resin does not introduce contaminants that could migrate into the cosmetic formula. At Oulete, we test PCR materials (PP, PE, and PET at ratios from 10% to 50%) through SGS-certified laboratories to verify material safety before production begins.
According to the FDA’s import guidance, all imported cosmetics are subject to the same FDA standards as domestically produced products. U.S. Customs and Border Protection examines cosmetic shipments at entry and can refuse entry for products that are adulterated or misbranded. “Misbranded” includes packaging with incorrect labeling, missing required information, or deceptive container design.
The practical workflow for a Chinese cosmetic packaging manufacturer exporting to U.S. brands involves several documentation steps. The manufacturer provides material safety data sheets, SGS or equivalent test reports, and detailed label artwork proofs. The U.S. brand or their importer of record ensures the final labeled product meets all FDA requirements before shipment.
Oulete produces over 20 million sets annually across 20 injection molding machines. Every production run for U.S.-bound packaging includes documentation of resin grade, colorant compliance (particularly for color additives that require FDA approval), and dimensional verification to ensure label area meets PDP requirements.
Stacking Compliance for Global Markets
FDA compliance is the U.S. layer, but most brands selling internationally need their packaging to satisfy multiple regulatory frameworks simultaneously. The physical packaging design must accommodate different label content requirements across markets without requiring separate tooling for each region.
EU REACH packaging requirements govern chemical substances in packaging materials sold in the European market. REACH restricts specific substances (such as certain phthalates and heavy metals) that may be present in plastic packaging. A packaging design that meets both FDA and EU REACH requirements gives brands a single SKU for two major markets.
Halal cosmetic packaging adds material-origin requirements. Certain animal-derived materials and alcohol-based coatings that are acceptable under FDA rules may disqualify packaging for halal certification. Organic certification packaging introduces its own material restrictions, limiting the types of plastics, coatings, and adhesives that can be used.
On our production floor, multi-market compliance stacking starts during the design phase. We map every required text element for each target market, calculate the total label surface area needed, and then recommend the container geometry that accommodates all requirements. For brands targeting the U.S., EU, and Middle East simultaneously, this often means selecting a container with a larger flat-panel area or using a wraparound label rather than direct printing.
According to Towards Packaging, the U.S. cosmetic packaging market alone is projected to grow from USD 2.1 billion in 2025 to USD 3.27 billion by 2034 at a CAGR of 5.05%. Brands that build multi-market compliant packaging now avoid retooling costs as they expand into new regions.
Practical Compliance Checklist for Brands
The following sequence is what we walk brands through when they are sourcing cosmetic packaging for the U.S. market. Each step addresses a specific regulatory requirement, and skipping any one of them can result in delays at the border.
Step 1: Map every required text element before selecting container size. List the product identity, net quantity, full INCI ingredient declaration, responsible person contact information, and any applicable warning statements. Calculate the total text area needed at FDA-compliant minimum type sizes. Only then select the container geometry that provides sufficient PDP and information panel area.
Step 2: Confirm your responsible person designation. The responsible person named on the label must have a U.S. domestic address, phone number, or electronic contact. For brands sourcing packaging from overseas, this is typically the U.S. importer or distributor, not the foreign manufacturer. This designation must be finalized before label artwork begins.
Step 3: Register your facility and list your products in the FDA database. If your facility manufactures or processes cosmetics for the U.S. market, MoCRA requires FDA registration. Product listing is a companion requirement with annual updates. Small businesses under $1 million in annual U.S. cosmetic sales may qualify for exemptions, but products used near the eye or intended for injection are excluded from this exemption.
Step 4: Request material safety documentation from your packaging supplier. This includes resin grade certificates, colorant compliance documentation (particularly for color additives requiring FDA approval), and migration testing results confirming the packaging does not contribute to product adulteration.
Step 5: Validate label artwork with your regulatory team before committing to print tooling. Label changes after printing plates or screens are cut add cost and delay. Pre-production proofing with regulatory review eliminates the most expensive compliance failure mode: relabeling finished inventory.
The brands that treat compliance as a packaging design input, rather than a last-minute label fix, consistently avoid the kind of customs holdups and relabeling costs that derail product launches. The packaging is the first thing regulators see, and it should be the first thing your supply chain gets right.
FAQ: FDA Cosmetic Packaging Compliance
Do cosmetics need FDA approval before they go on the market?
Cosmetic products do not require FDA pre-market approval. The manufacturer or distributor is responsible for ensuring the product is safe and properly labeled before sale. The one exception is color additives, which must receive FDA approval before use in any cosmetic product, according to the FDA.
What information is required on a cosmetic product label under FDA rules?
Every cosmetic label must display the product identity, net quantity of contents, ingredient declaration in descending order of predominance using INCI names, the name and place of business of the manufacturer or distributor, and (since December 2024) the responsible person’s domestic contact information. Warning statements are required for specific product categories.
What is MoCRA and how does it affect cosmetic packaging?
MoCRA (Modernization of Cosmetics Regulation Act of 2022) is the first major update to U.S. cosmetics law since 1938. It requires facility registration, product listing, adverse event reporting, and adds a mandatory responsible person contact information line to all cosmetic labels. It also introduces cGMP requirements for cosmetic manufacturers, though the FDA has delayed finalizing that rule.
Does FDA approve the packaging material used for cosmetics?
The FDA does not certify or approve specific packaging materials for cosmetics. However, the packaging must not cause the product to become adulterated. If container materials leach into or contaminate the product, the product violates federal law. Migration testing is the manufacturer’s responsibility.
How do imported cosmetics from China get cleared through U.S. Customs?
All imported cosmetics must comply with the same FDA standards as domestic products, according to the FDA. U.S. Customs and Border Protection examines shipments at entry. Products that are adulterated (unsafe) or misbranded (incorrect labeling) can be refused entry. The importer of record and the U.S.-based responsible person bear the compliance obligation.
What is the responsible person contact information requirement?
Since December 29, 2024, every cosmetic product label must include the responsible person’s domestic contact information, which can be a U.S. physical address, phone number, or electronic contact. The responsible person is the manufacturer, packer, or distributor named on the product label. This requirement was introduced by MoCRA.
What are the penalties for non-compliant cosmetic packaging?
The FDA can issue warning letters, seize products, seek injunctions, and refuse entry of imported products. MoCRA expanded FDA’s authority to include mandatory recall powers for cosmetics posing a serious health risk. Products found misbranded at the border can be detained, refused entry, or destroyed at the importer’s expense.
How does the FDA distinguish a cosmetic from a drug, and why does it matter for packaging?
A product is a drug if it claims to treat, cure, mitigate, or prevent disease, or if it affects the structure or function of the body. A product making both cosmetic and drug claims (such as an anti-acne moisturizer) must comply with both cosmetic and drug labeling requirements. This distinction directly affects what must appear on the packaging label.


